Organoid tumor models: benefits and challenges
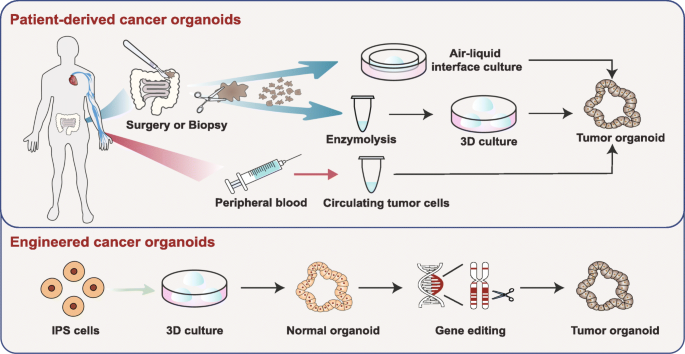
Cancer is one of the most threatening diseases and leading cause of death worldwide (1). Intra- and intertumoral heterogeneity is often responsible for metastasis formation, treatment failure, and therapy resistance. The increase in the cost of research and development of new anti-cancer therapeutics and the lack of translatability of pre-clinical cell-based assay and animal models to human patients have given a significant need to focus on personalized tumor models.
Tumor microenvironment and organoid technology
The tumor microenvironment is the complex environment in which tumor cells are embedded and comprises stromal cells, immune cells, signaling molecules, and the extracellular matrix (ECM). 3D organoid technology enables to recapitulate the structure, functionality, and molecular characteristics of the tumor microenvironment (2).
Tumoroids or ‘tumor-like organoids’ are derived from the original patient tumor and mimic the human tumor microenvironment. The inclusion of tumor microenvironment in personalized tumor models will serve as an important component to discover potential therapeutic targets (3).
Advantages of Tumoroids
– Tumoroids or 3D tumor models preserve the architectural and functional characteristics of native tissues.
– Cell differentiation and cell proliferation rates are in vivo-like.
– 3D organoids make the analysis of gene function and cell phenotype ex vivo more relevant.
– Tumoroids make it possible to study morphology and cellular changes shaped by altered ECM interactions during oncogenic transformation.
– 3D cell culture enhances the dimensionality of cell-cell interactions fundamental to generating a phenotype predictive of in vivo biology (4,5).
Challenges of Tumoroids
– Organoid cultivation requires condition optimization to produce in vivo-like phenotypic characteristics. Large-scale production of organoids is limited and this inhibits the application of organoids in high-throughput drug screening.
– In vitro organoids cannot completely recapitulate all the dynamic processes that occur in vivo such as blood circulation, thereby hindering the potential to accurately predict clinical outcomes.
– The generation of patient-derived organoids is expensive. (6).
References
1. World Health Organization cancer statistics
2. Finnberg NK, Gokare P, Lev A, Grivennikov SI, MacFarlane AW 4th, Campbell KS, Winters RM, Kaputa K, Farma JM, Abbas AE, Grasso L, Nicolaides NC, El-Deiry WS. Application of 3D tumoroid systems to define immune and cytotoxic therapeutic responses based on tumoroid and tissue slice culture molecular signatures. Oncotarget. 2017 Aug 5;8(40):66747-66757. doi: 10.18632/oncotarget.19965. PMID: 28977993; PMCID: PMC5620133.
3. Lee JY, Chaudhuri O. Modeling the tumor immune microenvironment for drug discovery using 3D culture. APL Bi9oeng. 2021 Feb 2;5(1):010903. doi: 10.1063/5.0030693. PMID: 33564739; PMCID: PMC7857858.
4. Kaushik G, Ponnusamy MP, Batra SK. Concise Review: Current Status of Three-Dimensional Organoids as Preclinical Models. Stem Cells. 2018 Sep;36(9):1329-1340. doi: 10.1002/stem.2852. Epub 2018 Jul 29. PMID: 29770526; PMCID: PMC6158106.
5. Habanjar O, Diab-Assaf M, Caldefie-Chezet F, Delort L. 3D Cell Culture Systems: Tumor Application, Advantages, and Disadvantages. Int J Mol Sci. 2021 Nov 11;22(22):12200. doi: 10.3390/ijms222212200. PMID: 34830082; PMCID: PMC8618305.
6. Xu H, Jiao D, Liu A, Wu K. Tumor organoids: applications in cancer modeling and potentials in precision medicine. J Hematol Oncol. 2022 May 12;15(1):58. doi: 10.1186/s13045-022-01278-4. PMID: 35551634; PMCID: PMC9103066.
