Cell Viability Testing Using Multicellular Tumor Spheroids (MCTs)
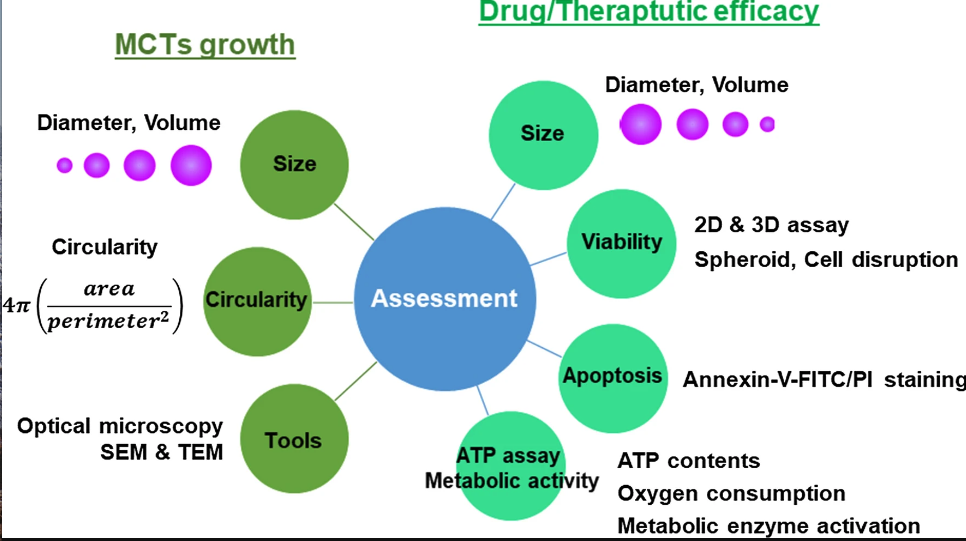
Cellular viability plays an important role when determining the efficacy and reliability of a MCTs model. There are many established viability assays such as colorimetric, luminescence and fluorescence assays. However these assays are well suited and optimized for traditional 2D cultures. There are a few assays developed for testing cell viability in 3D cell cultures that includes Cultrex® 3D colorimetric and CellTiter-Glo luminescence assays. However regardless of these commercial assays, most cell viability evaluations of MCTs still relies on those optimized for 2D cultures (1,2).
Conventional viability assays
Cell viability assays are determined via a fluorescence signal given by disintegrated cells or whole compact spheroids. Conventionally, MCTs are disrupted and cell lysate or suspension is analyzed. However this method of analysis do not account for multiple aspects of the spheroids, including any morphological changes. To avoid such poor readouts, imaging of intact MCTs are preferred, albeit being a challenging task caused by the prolonged image acquisition times and large and complex data processing involved (2).
Optimization of viability assays
There are several ways a conventional viability assay can be optimized for use in 3D cultures (1).
1) Improved incubation time with assay reagents depending on the size of spheroid. Adequate amount of time should be allowed for sufficient penetration of assay reagents.
2) Taking into account the compactness of the spheroid. Larger and more compact MCTs will require prolonged incubation times with assay reagents to ensure adequate penetration.
3) Fluorescence readout should be analyzed in consideration of the size and structural characteristics of the MCTs.
ATP and apoptosis assays
Cell death in MCTs is typically analyzed using flow cytometry by annexin V/PI staining. MCTs are disrupted in to a single cell suspension using enzymatic dissociation (3-5). It is important that the cells undergo a complete dissociation without altering their viability in order to get an accurate readout of apoptosis.
Cell viability of MCTs is also analyzed by measuring ATP content within the cells. The variable physical characteristics of MCTs such as size, composition, and penetration depth can affect the accuracy of the readouts, albeit recent attempts at optimizing the detergent composition and lysis conditions have proved effective at overcoming these challenges (6-8). Additionally ATP content is detected using bioluminescence, which incidentally offers robust, sensitive data scalable for high throughput screening. Other markers of cell viability that includes oxygen/nutrient consumption and metabolic enzyme activity are also routinely employed to assess cell viability in MCTs (6,9-10).
References
1. Vinci M, Gowan S, Boxall F, Patterson L, Zimmermann M, William C, et al. Advances in establishment and analysis of three-dimensional tumor spheroid-based functional assays for target validation and drug evaluation. BMC Biol. 2012;10(1):29
2. Zanoni M, Piccinini F, Arienti C, Zamagni A, Spartaco SS, Polico R, et al. 3D tumor spheroid models for in vitro therapeutic screening: a systematic approach to enhance the biological relevance of data obtained. Sci Rep. 2016;6:19103.
3. Gong X, Lin C, Cheng J, Su J, Zhao H, Tianlin L, et al. Generation of multicellular tumor spheroids with microwell-based agarose scafolds for drug testing. PLoS ONE. 2015;10(6):e0130348.
4. Khaitan D, Chandna S, Arya MB, Dwarakanath BS. Establishment and characterization of multicellular spheroids from a human glioma cell line; Implications for tumor therapy. J Transl Med. 2006;4:12.
5. Sanitini MT, Rainaldi G, Indovina PL. Apoptosis, cell adhesion and extracellular matrix in 3-D growth of multicelluler tumor spheroids. Critical Rev Oncol Hematol. 2001;36:75–87.
6. Kijanska M, Kelm J. In vitro 3D spheroids and microtissues: ATP-based cell viability and toxicity assays. Assay Guidance Manual. 2016;8:165.
7. Fey SJ, Wrzesinski K. Determination of drug toxicity using 3D spheroids constructed from an immortal human hepatocyte cell line. Toxicology Sciences. 2012;127(2):403–11.
8. Rimann M, Angres B, Patocchi-Tenzer I, Braum S, Graf-Hausner U. Automation of 3D cell culture using chemically defned hydrogels. J Lab Autom. 2014;19(2):191–7.
9. Thoma CR, Zimmermann M, Agarkova I, Kelm JM, Krek W. 3D cell culture systems modeling tumor growth determinants in cancer target discovery. Adv Drug Deliv Rev. 2014;69:29–41.
10. Emmert MY, Hitchcock RW, Hoerstrup SP. Cell therapy, 3D culture systems and tissue engineering for cardiac regeneration. Adv Drug Deliv Rev. 2014;69:254–69.
11. Han, S.J., Kwon, S. & Kim, K.S. Challenges of applying multicellular tumor spheroids in preclinical phase. Cancer Cell Int 21, 152 (2021). https://doi.org/10.1186/s12935-021-01853-8.
