The Role Of Cell Type In Multicellular Tumor Spheroid Formation
Multicellular tumor (MCT) spheroids have become a vital tool in advancing tumor research in pathophysiological front, as well as in developing effective therapeutics. However, its reliability and reproducibility is determined by several factors one of which is the uniformity of size and shape of the spheroids. The size and shape of 3D culture spheroids differ significantly from that of traditional 2D monolayer cultures. MCTs establish and maintain strong cell-cell, cell-extracellular matrix (ECM) connections, ultimately affecting spheroid formation. Apart from these connections, the cell type, culture techniques, medium compositions and cell density are other factors that can cause variability’s within the MCTs, thus affecting its overall reproducibility (1).
The structure of MCTs
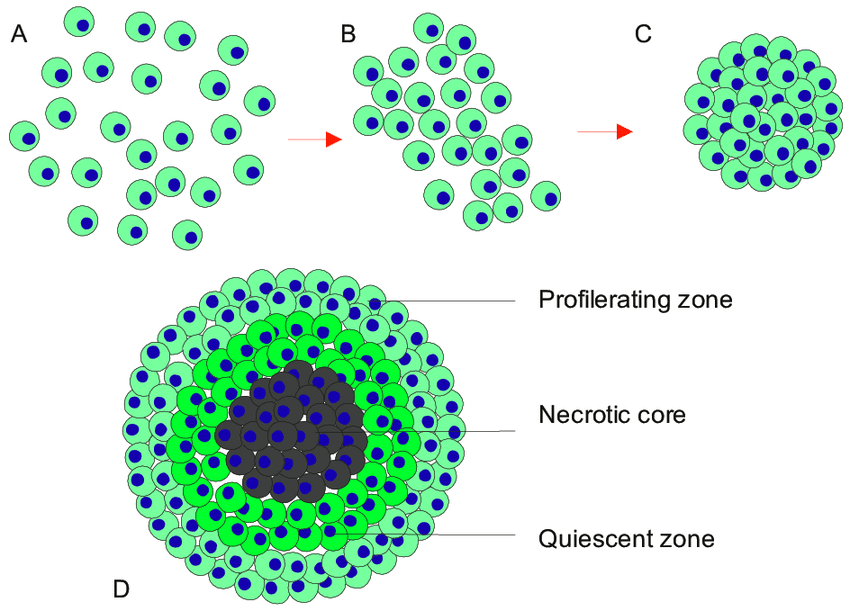
MCTs are primarily cultured with cancer cells, or co-cultured with other cell types such as fibroblasts, endothelial cells and immune cells. The cells typical aggregate and forms spherical spheroids merely days after seeding, encouraged by the use of low adhesion surfaces. The initial phase of aggregation is further facilitated by integrin-mediated adhesion to ECM and then compactly aggregated by E-cadherins. Once the spheroids have formed they contain a remarkable resemblance to in-vivo solid tumors, complete with a heterogeneous cell populations, oxygen and nutrient gradients. According to the oxygen and nutrient gradients present within the spheroid, MCTs exhibit a proliferating outer layer, quiescent cells present on the inner layer and a necrotic core (2-6).
MCT formation based on cell type
In the past decades many tumor cell types have been study to determine their ability to form reliable MCTs. Some cells show natural affinity for efficient spheroid formation, whereas others were found unsuccessful. Within the same cancer, the type of cell line used can also affect spheroid formation. For e.g. in breast cancer cells, the cell lines MCF-7, BT474, T47D and MDA-MB361 cell lines indicated compact spheroid formation, where as other breast cancer cell lines formed tight spheroids or loose aggregates which eventually disintegrated (7). Similarly gastric cancer cell lines such as RF-1, RF-48, and Hs-746T generated compact spheroids, while cell lines such as MKN-28, MKN-74 and N87 and SNU-5, SNU 6 cell lines formed tight and loose aggregates respectively.
Such variability is a result of innate differences within cell lines, in cell-cell adhesion, thus affecting the overall MCT formation. Cell lines with compact spheroid formation indicated high levels of E-cadherins and those with tight spheroid formation expressed high levels of N-cadherin molecules. Thus the formation of a compact spheroid suitable for a reliable MCT depends on the expression and type of cellular adhesion molecules and must be taken into account when choosing an appropriate cell line used for further study or drug screens (7,8).
References
1. Han, S.J., Kwon, S. & Kim, K.S. Challenges of applying multicellular tumor spheroids in preclinical phase. Cancer Cell Int 21, 152 (2021). https://doi.org/10.1186/s12935-021-01853-8
2. Courau T, Bonnereau J, Chicoteau J, Bottois H, Remark R, Laura AM, et al. Cocultures of human colorectal tumor spheroids with immune cells reveal the therapeutic potential of MICA/B and NKG2A targeting for cancer treatment. Journal for ImmunoTherapy of Cancer. 2019;7:74.
3. Rodríguez CE, Moverer LM, Reidel SI, Marino L, de Kier J, Maria AJ, et al. Cytotoxic efect of trastuzumab on macrophage-infltrated human mammary tumor spheroids. Cancer Res. 2012;72:2881.
4. McIntyre A, Patiar S, Wigfeld S, Li JL, Ledaki I, Helen T, et al. Carbonic anhydrase IX promotes tumor growth and necrosis in vivo and inhibition enhances anti-VEGF therapy. Clin Cancer Res. 2012;18:3100–11. 32.
5. Michele Z, Michela C, Alice Z, Chiara A, Sara P, Anna T. Modeling neoplastic disease with spheroids and organoids. J Hematol Oncol. 2020;13:97.
6. Hongxu L, Martina HS. Multicellular tumor spheroids (MCTS) as a 3D in vitro evaluation tool of nanoparticles. Small. 2018;14:1702858.
7. Ivanov DP, Grabowska AM. Spheroid arrays for high throughput singlecell analysis of spatial patterns and biomarker expression in 3D. Sci Rep. 2017;7:41160.
8. Mayer B, Klement G, Kaneko M, Man S, Jothy S, Rak J, et al. Multicellular gastric cancer spheroids recapitulate growth pattern and diferentiation phenotype of human gastric carcinomas. Gastroenterology. 2001;121(4):839–52.
9. Kamatar A, Gunay G, Acar H. Natural and Synthetic Biomaterials for Engineering Multicellular Tumor Spheroids. Polymers. 2020; 12(11):2506.
