Perfect 3D spheroids for cancer research with BIOFLOAT
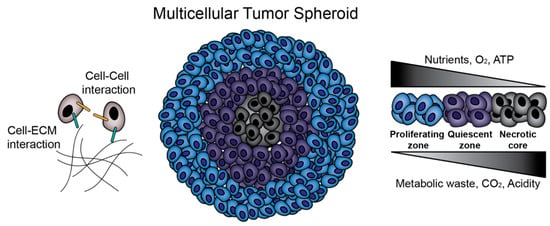
Cancer research relies on cell based model systems to understand the growth and progression of tumors, and subsequently the discovery of new therapeutic drugs. Traditional monolayer cultures have been widely used in this regard due to its ability to generate well-controlled homogenous cell environment and ease of microscope analysis. However 2D cultures are unable to recapitulate the in-vivo microenvironment of the cells and as a result most drug screenings done on 2D monolayer cells falls short when transferred to animal models or humans (1).
3D cell culture models have become the method of choice to overcome the limitations found on monolayer cells. In a 3D cell culture platform, tumor cells are grown in suspension, and self aggregate in to 3D spheroids consisting of extracellular matrix (ECM) (2). These aggregates then mimic in-vivo like cell-cell and cell-ECM interactions in each dimension, and activation of cellular pathways typical of solid tumors. The dense 3D network also acts as a physical barrier for drugs and includes high number of quiescent cells with an increased survival rate and cancer-specific gene expression (2,3). The conditions are ideal for creating a tumor microenvironment in vivo. Additionally tumor spheroids are found to be more resistant to chemotherapeutic drugs making them applicable for use in drug screens (3).
Spheroids are commonly cultured in well plates coated with special coating such as BIOFLOAT Flex coating solution. These coating ensures a hydrophilic cell repellant coating forcing the cells to self-aggregate. Spheroid systems can be modified by; i) spheroid morphology, ii) spheroid number, iii) spheroid formation over time, iv) spheroid size, and v) cell line and co-culture. All of these parameters can determine the reliability and reproducibility of these cultures (4, 5).
To this end, BIOFLOAT Flex or pre coated plates offer the following advantages:
1) When compared with benchmark plates, BIOFLOAT coated plates allow the formation of one round cell spheroids per well, which is an important criterion for generating a reproducible, reliable cell and high-throughput analysis, as such analysis require an even distribution of one (symmetrical) spheroid per well for the specific readout by automated systems (5).
2) In most cell culture systems the size of spheroid is determined by the initial seeding density, which can differ between cell lines.
3) To recapitulate the cellular heterogeneity found in solid tumors, spheroids are often cultured in co-cultures of different ratios of cancer to stromal cells to better mimic the in vivo conditions Most often, one cell line acts as feeder cells for the other cell line (5, 6).
For cancer research it is of importance to generate a modifiable in vitro platform to understand the growth and progression of solid tumors. In this regard, BIOFLOAT spheroid
culture system is an excellent choice that allows generation of round and uniform spheroids, in which spheroid morphology can be fine-tuned easily by modifying initial seeding number or cell line model. Furthermore co-cultures of tumor cells with multiple cell lines can be achieved to build up complex and relevant models for fundamental cancer or pharmaceutical research.
References
1. Jensen, C. and Y. Teng, Is It Time to Start Transitioning From 2D to 3D Cell Culture? Front Mol Biosci, 2020. 7(33).
2. Costa, E.C., et al., 3D tumor spheroids: an overview on the tools and techniques used for their analysis. Biotechnol Adv, 2016. 34(8): p. 1427-1441.
3. Imamura, Y., et al., Comparison of 2D- and 3D-culture models as drug-testing platforms in breast cancer. Oncol Rep, 2015. 33(4): p. 1837-43.
4. Costa, E.C., et al., Optimization of liquid overlay technique to formulate heterogenic 3D co-cultures models. Biotechnol Bioeng, 2014. 111(8): p. 1672- 1685. 5. https://facellitate.com/wp-content/uploads/Establishment-of-perfect-3D-spheroids-for-1.pdf
6. Kamatar A, Gunay G, Acar H. Natural and Synthetic Biomaterials for Engineering Multicellular Tumor Spheroids. Polymers. 2020; 12(11):250
