Application Notes
Three-dimensional (3D) spheroids provide a more physiologically relevant cancer model than conventional 2D cultures, as they better mimic tumor architecture, cell–cell interactions, and drug resistance. The formation of uniform, round spheroids is essential for reproducible results and reliable high-throughput screening. Spheroid size and density are mainly influenced by the cell line and the initial seeding density, and these parameters must be optimized for each model. In addition, coculture spheroids incorporating stromal or endothelial cells more closely resemble the in vivo tumor microenvironment. Together, these considerations form a practical guideline for establishing robust and translational 3D spheroid models.
Three-dimensional (3D) astrocyte spheroid models provide a reliable and human-relevant platform to study brain infections, neurotoxicity, and neurodegenerative diseases. Compared to 2D cultures, 3D astrospheroids better mimic the dense cellular architecture, cell–cell interactions, and functional characteristics of astrocytes in vivo. Using low-attachment surfaces enables rapid formation of compact, round, and uniform astrocyte spheroids with high viability and consistent morphology. Such standardized spheroid models are particularly well suited for reproducible readouts in drug testing and toxicological screening. Overall, 3D astrocyte spheroids help bridge the gap between animal models and human brain physiology.
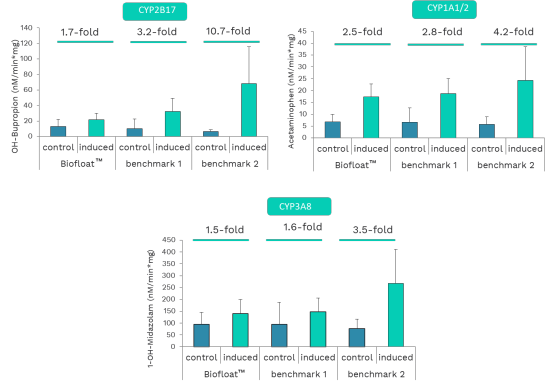
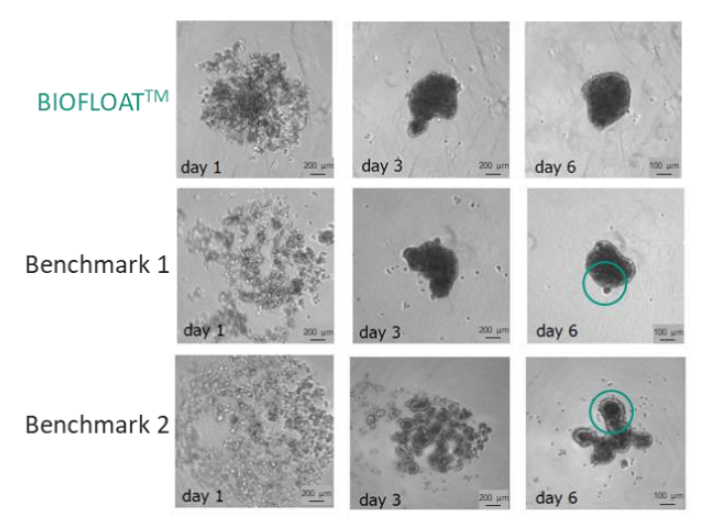
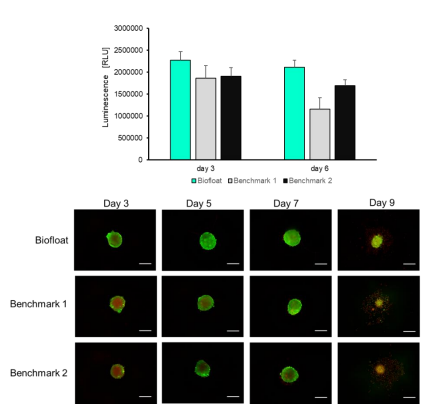
Uniform 3D hepatocyte spheroids offer a more physiologically relevant model than 2D cultures, preserving key liver-specific functions such as cytochrome P450 activity. This study demonstrates that ultra-low attachment surfaces generated with a novel polymer solution enable rapid and reproducible formation of compact, round spheroids from primary hepatocytes. Compared to benchmark low-attachment plates, BIOFLOAT™ surfaces produced more homogeneous spheroids with well-defined geometry and consistent formation kinetics. Importantly, spheroids formed on these fully inert surfaces showed robust viability and controlled drug accessibility, supporting reliable functional readouts. Overall, careful selection of the culture surface is critical for generating high-quality primary hepatocyte spheroids suitable for routine testing and translational research.
