Non-Adhesive Well Plates For Spheroid Generation
Spheroid cell cultures are defined as 3-dimentional cell aggregates that are formed via spontaneous self-assembly of cells in a cell repellant, non-adhesive surface. It is by far one of the popular anchorage free 3D cell culture methods, that are low tech and low cost, compared to other conventional 3D culture platforms.
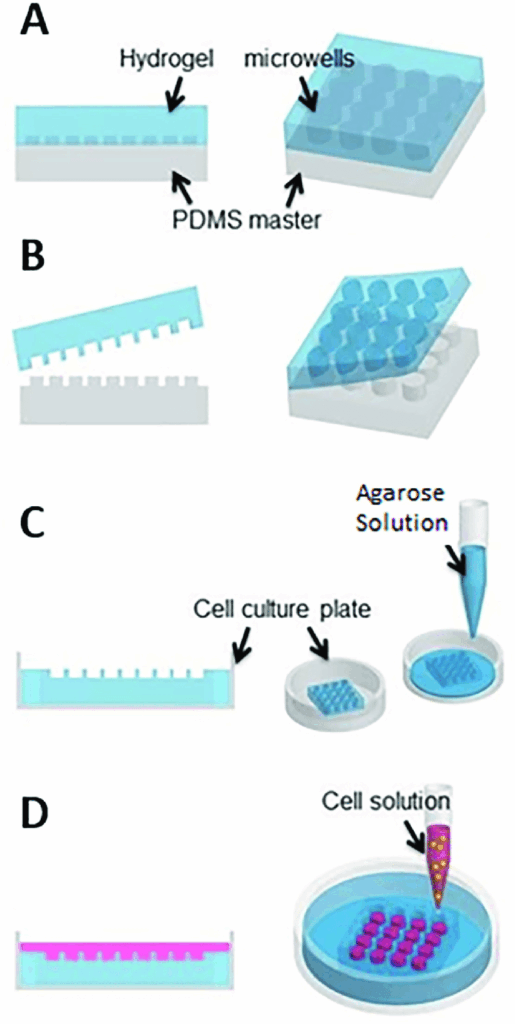
Creating Non adhesive surfaces
The non-adhesive surfaces used to form spheroids are called a liquid overlay, and facilitates the formation of spheroids by preventing protein adsorption and cell attachments thus allowing cell-cell aggregation (1).
Agar and agarose are popular biomaterials used to modify non-adhesive surfaces. Both agar and agarose are relatively low cost, is soluble at high temperatures of 650C or higher and are compatible with autoclave-based sterilization. However despite its utility, agar or agarose coated plates remain usable only for a few days, and a limited usage time (2-4).
An alternative to agar and agarose is poly (2-hydroxyethyl methacrylate) (poly-HEMA). This particular coating material is more expensive, but offers the advantage extended storage time, and longer usage period over several months. However using poly-HEMA to modify a non-adhesive surface requires 5-7hours, with added additional time to evaporate the ethanol used as a solvent. Although it is more stable over a long period than agar or agarose, the process is costly and time consuming and thus affects, reproducibility (2,5).
At the beginning non-adhesive surfaces were made using petri dishes or cell culture flasks. However spheroids on flat, and wider surface tends to be variable in size (2). In the recent times, round-bottomed 96 well plates are used to form spheroids. In these plates the cells aggregate at the bottom of the wells via gravity. When compared with other conventional spheroid culture methods such as the hanging drop method, these cultures are easier to handle and requires little to no external manipulation. One of the biggest advantages is the ability to control spheroid size. As all the cells are put in to one well to form one spheroid, regulating the number of cells in the cell suspension can regulate the size of the spheroid (6).
Commercialized non adhesive well plates
Presently there are many commercialized coated plates available that are highly effective across a wide range of cell lines. These include U bottom microplates with treated non-adhesive surfaces and BiofloatTM well plates coated with special polymeric coating, that renders the surface cell repellant. BiofloatTM Flex solution can also be used to modify other cell culture surfaces into non-adhesive surfaces. The commercialized products carry the advantage of improving end-user convenience in terms of reducing spheroid formation time, thus contributing to increased uniformity and reproducibility. However, microplates with single spheroid per well still remains incompatible with large-scale spheroid production. Regardless, microplates with modified non-adhesive surface is by far the simplest method to generate uniform spheroids in a time and cost effective manner (7,8).
References
1. Kim, S.J., Kim, E.M., Yamamoto, M., Park, H., Shin, H.: Engineering Multi-Cellular Spheroids for Tissue Engineering and Regenerative Medicine. Adv. Healthc. Mater. 9, e2000608 (2020).
2. Costa, E.C., de Melo-Diogo, D., Moreira, A.F., Carvalho, M.P., Correia, I.J.: Spheroids formation on non-adhesive surfaces by liquid overlay technique: considerations and practical approaches. Biotechnol. J. 13, 1700417 (2018).
3. Dalen, H., Burki, H.: Some observations on the three-dimensional growth of L5178Y cell colonies in soft agar culture. Exp. Cell Res. 65, 433–438 (1971).
4. Su, G., Zhao, Y., Wei, J., Han, J., Chen, L., Xiao, Z., Chen, B., Dai, J.: The efect of forced growth of cells into 3D spheres using low attachment surfaces on the acquisition of stemness properties. Biomaterials 34, 3215–3222 (2013).
5. Kuroda, Y., Wakao, S., Kitada, M., Murakami, T., Nojima, M., Dezawa, M.: Isolation, culture and evaluation of multilineagedifferentiating stress-enduring (Muse) cells. Nat. Protoc. 8, 1391–1415 (2013).
6. Pereira, P.M.R., Berisha, N., Bhupathiraju, N., Fernandes, R., Tome, J.P.C., Drain, C.M.: Cancer cell spheroids are a better screen for the photodynamic efciency of glycosylated photosensitizers. PLoS ONE 12, e0177737 (2017).
7. Park, S.Y., Hong, H.J. & Lee, H.J. Fabrication of Cell Spheroids for 3D Cell Culture and Biomedical Applications. BioChip J 17, 24–43 (2023). 8. https://facellitate.com/product-category/biofloat-3d-cell-culture/
