Mammospheres leading the way in breast cancer research
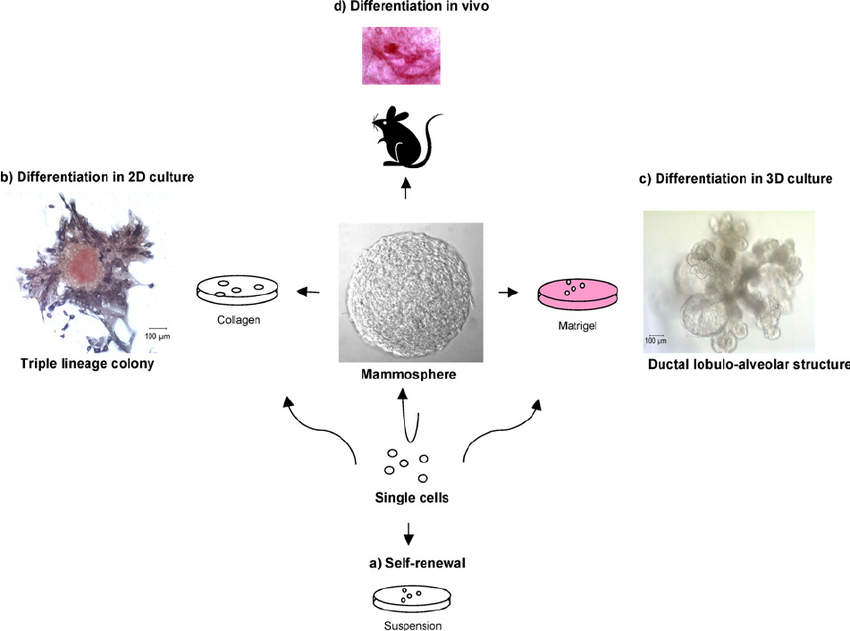
The advances in spheroid technology in particular, neurospheres has led to establishment of “mammospheres”; a mammalian cancer spheroid derived from human mammary epithelial cells. This has been an important milestone for breast cancer research as the presence of potential breast cancer stem cells are known to be resistant to conventional therapeutic measures available (1).
Culturing breast cancer spheroids
The mammospheres were developed enriched for early progenitor cells, and are capable of differentiating into the 3 primary mammary cell lineages. Once the cancer stem cells were identified (using markers such as CD24, CD44) (6) and sorted, traditional spheroid protocols were successfully employed to develop long term 3D cultures from breast cancer (2-6). These cells were found to express stem cell markers and formed xenograft tumours in immunocompromised mice, thus affirming the validity and reliability of the 3D culture model (3).
Reliability of mammospheres as an appropriate in vitro culture model
Such mammalian breast cancer spheroids have been successfully used to investigate intertumor heterogeneity, as well as to understand the proliferative capacity of IL-8/CXC motif chemokine receptor 1/2 mediated pathways and insulins-like growth factor 2 pathway. In a more clinically translatable context, mammospheres were also used to identify effects of chemical compounds on cancer stem cells. Mouse mammospheres are also used as a reliable platform to investigate other tumor related effects such as role of Erb-B2 receptor tyrosine kinase 2 expression, p53 deficiency along with the role of pathways such as Wnt/beta-catenin signalling pathway and Sox2 expression.

In essence the available reports suggest that the 3D mammosphere assay of metastatic breast cancer cells model proves to be a reliable, and representative model for studies involving characterisation of the tumour-inducing subpopulation of breast cancer cells as well as studying the sensitivity of cancer stem cells to therapeutic compounds (7).
References
1. Dontu G, Abdallah WM, Foley JM et al. In vitro propagation and transcriptional profiling of human mammary stem/progenitor cells. Genes Dev 2003;17: 1253–70.
2. Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA 2003;100: 3983–8.
3. Ponti D, Costa A, Zaffaroni Net al. Isolation and in vitro propagation oftumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res 2005; 65: 5506–11.
4. Jiao X, Rizvanov AA, Cristofanilli M, Miftakhova RR, Pestell RG. Breast cancer stem cell isolation. Methods Mol Biol 2016;1406: 121–35.
5. Shaw FL, Harrison H, Spence K et al. A detailed mammosphere assay protocol for the quantification of breast stem cell activity. J Mammary Gland Biol Neoplasia 2012;17: 111–6
6. Grimshaw, M.J., Cooper, L., Papazisis, K. et al. Mammosphere culture of metastatic breast cancer cells enriches for tumorigenic breast cancer cells. Breast Cancer Res 2008 10, R52.
7. Ishiguro T, Ohata H, Sato A, Yamawaki K, Enomoto T, Okamoto K. (2017). Tumor-derived spheroids: Relevance to cancer stem cells and clinical applications. Cancer Sci. 108(3):283-289.
8. Dontu, Gabriela & Wicha, Max. (2005). Survival of Mammary Stem Cells in Suspension Culture: Implications for Stem Cell Biology and Neoplasia. Journal of mammary gland biology and neoplasia. 10. 75-86.
