Homotypic and heterotypic spheroids
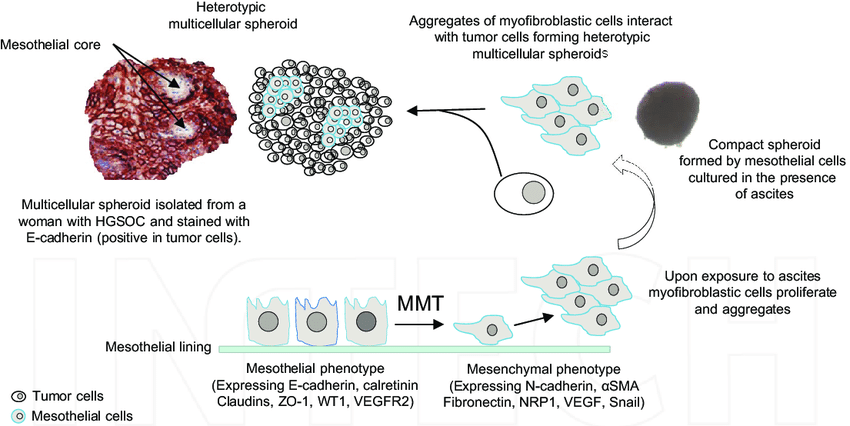
Solid tumors are composed of a number of different types of cells. These include cancer cells, stromal cells such as fibroblasts, immune cells, lymphatic endothelial cells, vascular endothelial cells, pericytes and adipocytes (1). These cells by it self are not harmful but cellular interaction between these cells and cancer cells are essential for tumor angiogenesis, proliferation, invasion and metastasis, and developing drug resistance mechanisms (2). Therefore 3D tumor spheroid modeling methods are constantly improved to better mimic these cell-cell interactions, by co-culturing cancer cells with stromal cells, endothelial cells or immune cells (3-5).
Homotypic and heterotypic spheroids
Homotypic spheroids are spheroids composed of a single type of cells, whereas heterotypic spheroids are consist of multiple cell types. The latter is grown in conventional media supplemented with serum, under non-adherent conditions (6).
Direct communications between stromal cells and cancer cells, together with cytokine, extracellular vesicle and growth factor release is crucial to recreate the complex signaling network found in in vivo solid tumors. There has been number of studies on different stromal: cancer cells ratios carried out to accurately recapitulate the specific tissue composition found in-vivo. For example fibroblasts are among the most abundant stromal cell populations in the tumor microenvironment that plays a significant role in tumor initiation, progression, metastasis and drug response. Thus heterotypic tumor spheroids are widely regarded as a reliable tumor model for drug discovery and tumor pathophysiological studies (6-10).
References
1. Balkwill FR, Capasso M, Hagemann T. The tumor microenvironment at a glance. J Cell Sci. 2012;125:5591–6.
2. Correia AL, Bissell MJ. The tumor microenvironment is a dominant force in multidrug resistance. Drug Resist Updat. 2012;15:39–49
3. Lao Z, Kelly CJ, Yang X-Y, Jenkins WT, Toorens E, Ganguly T, et al. Improved methods to generate spheroid cultures from tumor cells, tumor cells & fibroblasts or tumor fragments: Microenvironment. Microvesicles and MiRNA. PLoS One. 2015;10:e0133895.
4. Lamichhane SP, Arya N, Kohler E, Xiang S, Christensen J, Shastri VP. Recapitulating epithelial tumor microenvironment in vitro using three dimensional tri-culture of human epithelial, endothelial, and mesenchymal cells. BMC Cancer. 2016;16:581.
5. Rodríguez CE, Moverer LM, Reidel SI, Marino L, de Kier Joffé EDB, Jasnis MA, et al. Abstract 2881: cytotoxic effect of trastuzumab on macrophageinfiltrated human mammary tumor spheroids. Cancer Res. American Association for Cancer Research (AACR). 2012;72:2881–1.
6. Majety M, Pradel LP, Gies M, Ries CH. Fibroblasts influence survival and therapeutic response in a 3D co-culture model. PLoS One. 2015;10: e0127948.
7. McMillin DW, Negri JM, Mitsiades CS. The role of tumour-stromal interactions in modifying drug response: challenges and opportunities. Nat Rev Drug Discov. 2013;12:217–28.
8. Lee J-H, Kim S-K, Khawar IA, Jeong S-Y, Chung S, Kuh H-J. Microfluidic coculture of pancreatic tumor spheroids with stellate cells as a novel 3D model for investigation of stroma-mediated cell motility and drug resistance. J Exp Clin Cancer Res. 2018;37:4.
9. Hoffmann OI, Ilmberger C, Magosch S, Joka M, Jauch K-W, Mayer B. Impact of the spheroid model complexity on drug response. J Biotechnol. 2015;205:14–23
10. Matte, Isabelle & Bessette, Paul & Piché, Alain. (2017). Ascites in Ovarian Cancer Progression: Opportunities for Biomarker Discovery and New Avenues for Targeted Therapies. 10.5772/intechopen.70993.
