Applications of Multicellular Spheroids (MCS)
Multicellular spheroids are formed through spontaneous aggregation of cells in the presence of adhesion molecules known as cadherins (1). Since its first implementation in 1952 (2), multicellular spheroids have served as an efficient 3D cell culture model for a wide array of research. The methods for generating MCS are evaluated and standardized based on several parameters such as efficiency of formation, convenience, cost, and spheroid size (3).
Most traditional methods for forming MCS include liquid overlay method, including the microwells array method, the hanging drop method, rotating flasks, and the external force method. While these methods have been widely employed to generate MCS, they lack production efficiency, require intense labor, and fail in controlling spheroid size. Recent technological developments in micro/nanofabrication, cell imaging, and optics have paved the way for novel MCS generating methods that are able to form MCS compatible for high throughput assays while providing a more comprehensive view of inter/intra cellular processes involved in MCS formation. (3).
Applications of MCS
1. Tumor research
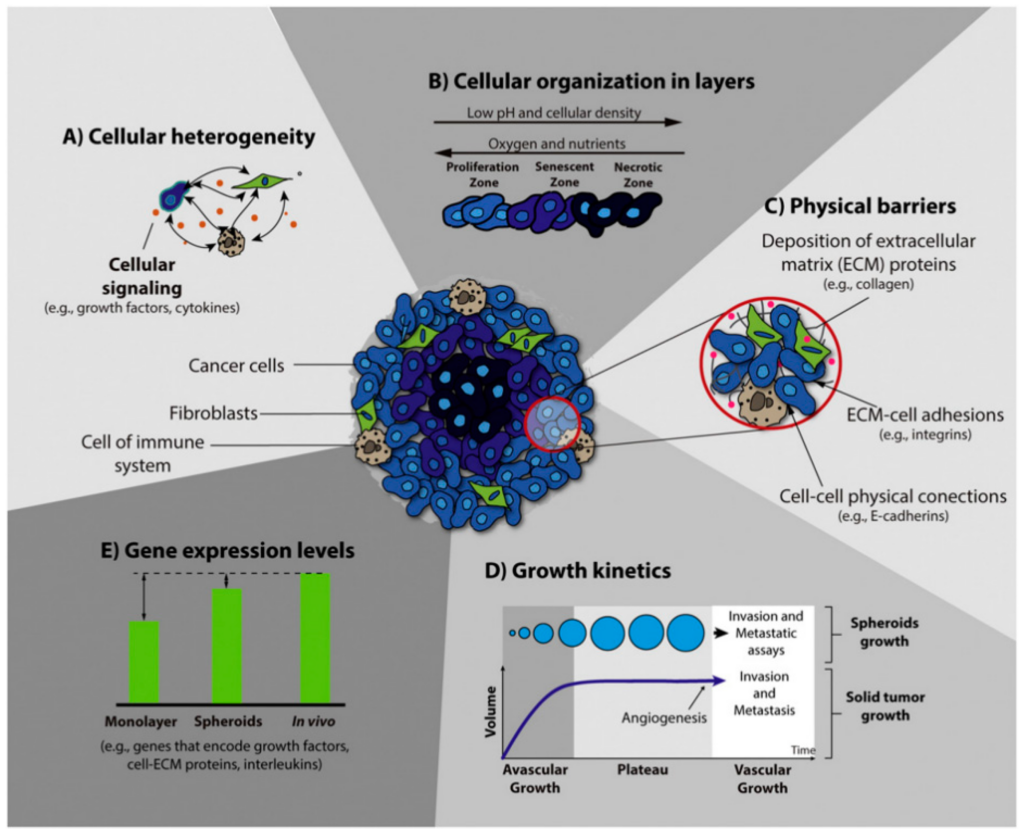
MCS are an ideal model for tumor research as they enable the co-cultivation of multiple cell types to form heterogeneous spheroids that mirror in vivo like cell-cell and cell-ECM interactions. Typically, tumor cells that are co-cultured with fibroblasts and endothelial cells replicate the in vivo-like complex cell environment. These MCS are able to indicate tumor specific cell behaviors such as angiogenesis, cell invasion, and metastasis (4-6).
2. In vitro drug screenings
Drug screenings are a vital step in the development of new medicaments that must be performed before a drug reaches the clinical trial phase. Traditionally, drug screening have been performed on 2D cell culture models. While still useful, these models lead to poor efficacy in trials, as they both lack cell-cell and cell-ECM interactions. The MCS model has thus proved to be ideal for drug screening, particularly for anti-tumor drugs. Drugs such as doxorubicin, cisplatin, and paclitaxel are some examples for which MCS has been successfully employed for investigating drug resistance and sensitivity (3,8).
3. Tissue engineering
Tissue engineering can be implemented in regenerative medicine to construct tissues or organs and replace damaged ones. Hydrogels are routinely used for this purpose as a scaffold due to their compatibility and biodegradability (9).
4. Anti-tumor response of immune cells
Anti tumor activity of immune cells is largely mediated by the T cells and Natural Killer (NK) cells. MCS cultures are used in cancer immunotherapy studies specifically to study the function and efficiency of the immune response to the presence of tumor cells (10).
References
1. 1. Lin, R.Z.; Chou, L.F.; Chien, C.C.; Chang, H.Y. Dynamic analysis of hepatoma spheroid formation: Roles of E-cadherin and beta1-integrin. Cell Tissue Res. 2006, 324, 411–422.
2. Moscona, A.; Moscona, H. The dissociation and aggregation of cells from organ rudiments of the early chick embryo. J. Anat. 1952, 86, 287–301.
3. Shen, H.; Cai, S.; Wu, C.; Yang, W.; Yu, H.; Liu, L. Recent Advances in Three-Dimensional Multicellular Spheroid Culture and Future Development. Micromachines 2021, 12, 96.
4. Noel, P.; Munoz, R.; Rogers, G.W.; Neilson, A.; Von Hoff, D.D.; Han, H. Preparation and Metabolic Assay of 3-dimensional Spheroid Co-cultures of Pancreatic Cancer Cells and Fibroblasts. J. Vis. Exp. 2017, 56081.
5. Chiew, G.G.Y.; Wei, N.; Sultania, S.; Lim, S.; Luo, K.Q. Bioengineered three-dimensional co-culture of cancer cells and endothelial cells: A model system for dual analysis of tumor growth and angiogenesis. Biotechnol. Bioeng. 2017, 114, 1865–1877.
6. Kim, S.A.; Lee, E.K.; Kuh, H.J. Co-culture of 3D tumor spheroids with fibroblasts as a model for epithelial-mesenchymal transition in vitro. Exp. Cell. Res. 2015, 335, 187–196.
7. Neto, A.I.; Correia, C.R.; Oliveira, M.B.; Rial-Hermida, M.I.; Alvarez-Lorenzo, C.; Reis, R.L.; Mano, J.F. A novel hanging spherical drop system for the generation of cellular spheroids and high throughput combinatorial drug screening. Biomater. Sci. 2015, 3, 581–585.
8. Akay, M.; Nguyen, D.T.; Fan, Y.; Akay, Y.M. Engineering a Three-Dimensional In Vitro Drug Testing Platform for Glioblastoma. J. Nanotechnol. Eng. Med. 2015, 6
9. Huang, C.C.; Chen, D.Y.; Wei, H.J.; Lin, K.J.; Wu, C.T.; Lee, T.Y.; Hu, H.Y.; Hwang, S.M.; Chang, Y.; Sung, H.W. Hypoxia-induced therapeutic neovascularization in a mouse model of an ischemic limb using cell aggregates composed of HUVECs and cbMSCs. Biomaterials 2013, 34, 9441–9450.
10. Courau, T.; Bonnereau, J.; Chicoteau, J.; Bottois, H.; Remark, R.; Assante Miranda, L.; Toubert, A.; Blery, M.; Aparicio, T.; Allez, M.; et al. Cocultures of human colorectal tumor spheroids with immune cells reveal the therapeutic potential of MICA/B and NKG2A targeting for cancer treatment. J. Immunother. Cancer 2019, 7, 74
