Spheroid culture techniques
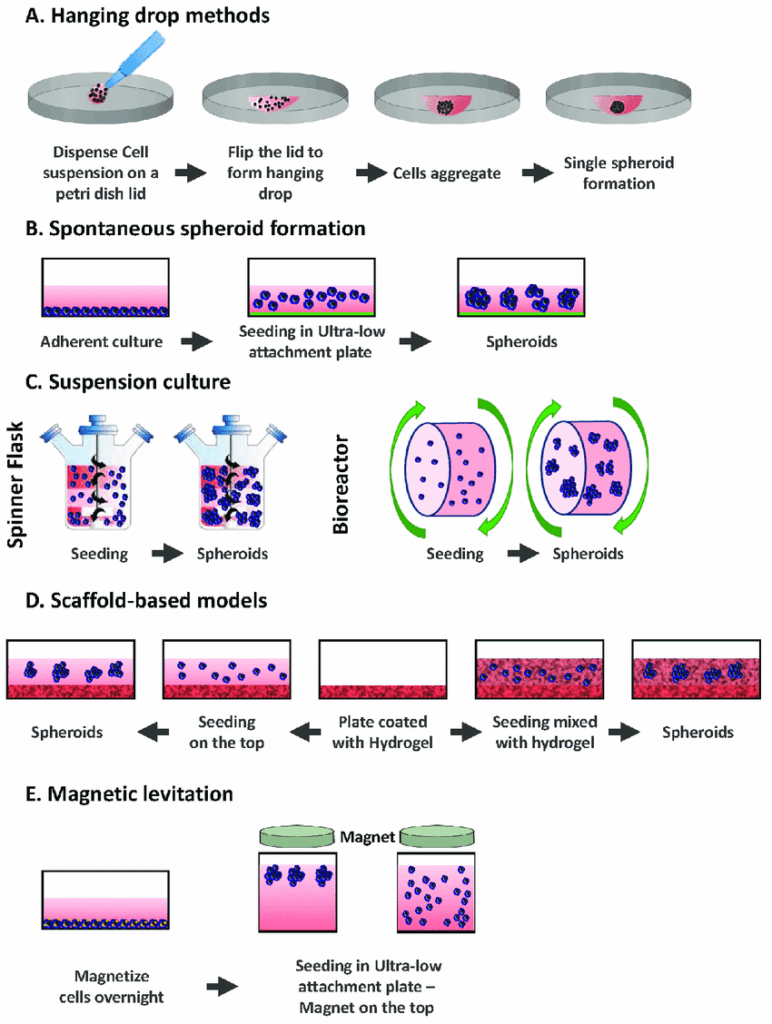
This article briefly describes the modern spheroid culturing techniques available.
Pellet cultures
In pellet culture systems, cells are forced to the bottom of the tube via centrifugation. Cell-cell adhesions are thus facilitated by proximity of single cells at the bottom. Supernatants are then removed, and the cell pellet resuspended in culture medium appropriate for spheroid formation. Once cell count is known, cell suspension is then added to individual wells of a 96-well U bottom plate with a cell repellant surface such as BIOFLOATTM 96 well plates. This method forms spheroids in a rapid, and reproducible platform, making it an excellent choice for high throughput drug screens (1-2).
Static suspension culture
In this technique, spheroids are formed by disrupting cell-cell adhesion on non-adherent flat bottom plates. These culture layers consist of agar or agarose gel. Agarose gel is the preferred option of liquid overlay method, as it efficiently prevents cell adhesion compared to agar. Disruption of cell adhesion in this manner then promotes spontaneous cell aggregation above the non-adherent surface. Alternative non-adherent biomaterial can be used such as hyaluronic acid, which incidentally is more suitable for use in tumor spheroid cultures (3).
Hanging drop method
Hanging drop culture technique involves single cell aggregation and spheroid in the form of droplets. The unique droplet shape is achieved by regulating the volume of the drop or density of cell suspension. This method enables to efficiently form fixed size spheroids. The general mechanics of hanging drop method comprises of a monolayer cell culture, which is prepared as a suspension and diluted with appropriate culture medium to attain the desired cell density. Then the cell suspension is added into wells of a mini-tray with the help of a compatible multichannel pipette. A lid is then placed on the mini-tray and the entire mini-tray is turned upside down. The cell suspension drops attached on the mini-tray would then remain on the reversed side by surface tension. Droplet spheroid formation is facilitated by concurrent action of surface tension and gravitational force (1,4).
Spinner Culture
This spheroid culture technique involves continuous stirring of the cell suspension in spinner flask bioreactor container. The resulting spheroid is controlled by size of the bioreactor container. Convectional force of the stirring bar determines the fluid and mass in the containers and consequently the formation of spheroids. If the cell suspension is stirred too fast, the cells may get damaged, whereas if stirred too slowly, the cells may sink to the bottom of the container preventing the formation of spheroids (1).
Rotating wall vessel

Rotating wall vessel technique recreates microgravity by constant circular rotation. As the cells are constantly rotated, they remain in suspension in the vessel, resulting in the formation of circular spheroids (5-6).
Lab on a chip
Lab on a chip is a microfluidic culture technique commonly used for single cell analysis, and drug toxicity screens. Lab on chip technique consists of microscale dimensions representative of in-vivo environment and thus recreating the native 3D structure. Microfluidic devices also include materials that easily permit oxygen and other growth factors essential for proliferation. This helps to decrease hypoxia, which is a common disadvantage associated with spheroid cultures. Modern fluidic devices also offer diverse designs, at a reduced cost, and allows real time monitoring of cells in a easily modifiable microenvironment, which makes them the prime choice for high throughout assays (7,8).
Magnetic levitation
Magnetic levitation system involves mixing the cells with magnetic particles and subjecting them to a magnetic force during culturing process. It utilizes negative magnetophoresis, which mimics a weightlessness state. The magnetic force exerted causes the cells combined with magnetic particles to stay levitated or floating against gravity. This condition causes a geometry change of cell mass and stimulates cellular contact, leading to cell aggregation. Additionally this system can also facilitate multi-cellular co-culturing with different cell types (9-11).
References
1. Achilli, T.M.; Meyer, J.; Morgan, J.R. Advances in the formation, use and understanding of multi-cellular spheroids. Expert Opin. Biol. Ther. 2012, 12, 1347–1360.
2. Maritan, S.M.; Lian, E.Y.; Mulligan, L.M. An efficient and flexible cell aggregation method for 3D spheroid production. J. Vis. Exp. 2017.
3. Costa, E.C.; de Melo-Diogo, D.; Moreira, A.F.; Carvalho, M.P.; Correia, I.J. Spheroids formation on non-adhesive surfaces by liquid overlay technique: Considerations and practical approaches. Biotechnol. J. 2018, 13.
4. Timmins, N.E.; Nielsen, L.K. Generation of multicellular tumor spheroids by the hanging-drop method. Methods Mol. Med. 2007, 140, 141–151.
5. Carpenedo, R.L.; Sargent, C.Y.; McDevitt, T.C. Rotary suspension culture enhances the efficiency, yield, and homogeneity of embryoid body differentiation. Stem Cells 2007, 25, 2224–2234.
6. Manley, P.; Lelkes, P.I. A novel real-time system to monitor cell aggregation and trajectories in rotating wall vessel bioreactors. J. Biotechnol. 2006, 125, 416–424.
7. Ziolkowska, K.; Jedrych, E.; Kwapiszewski, R.; Lopacinska, J.; Skolimowski, M.; Chudy, M. PDMS/glass microfluidic cell culture system for cytotoxicity tests and cells passage. Sens. Actuators B Chem. 2010, 145, 533–542.
8. Nie, F.Q.; Yamada, M.; Kobayashi, J.; Yamato, M.; Kikuchi, A.; Okano, T. On-chip cell migration assay using microfluidic channels. Biomaterials 2007, 28, 4017–4022.
9. Anil-Inevi, M.; Yaman, S.; Yildiz, A.A.; Mese, G.; Yalcin-Ozuysal, O.; Tekin, H.C.; Ozcivici, E. Biofabrication of in situ self assembled 3D cell cultures in a weightlessness environment generated using magnetic levitation. Sci. Rep. 2018, 8, 7239.
10. Kim, J.A.; Choi, J.H.; Kim, M.; Rhee, W.J.; Son, B.; Jung, H.K.; Park, T.H. High-throughput generation of spheroids using magnetic nanoparticles for three-dimensional cell culture. Biomaterials 2013, 34, 8555–8563.
11. Lewis, N.S.; Lewis, E.E.; Mullin, M.; Wheadon, H.; Dalby, M.J.; Berry, C.C. Magnetically levitated mesenchymal stem cell spheroids cultured with a collagen gel maintain phenotype and quiescence. J. Tissue Eng. 2017, 8, 204173141770442.
