Scaffold dependent 3D models for high throughput screening
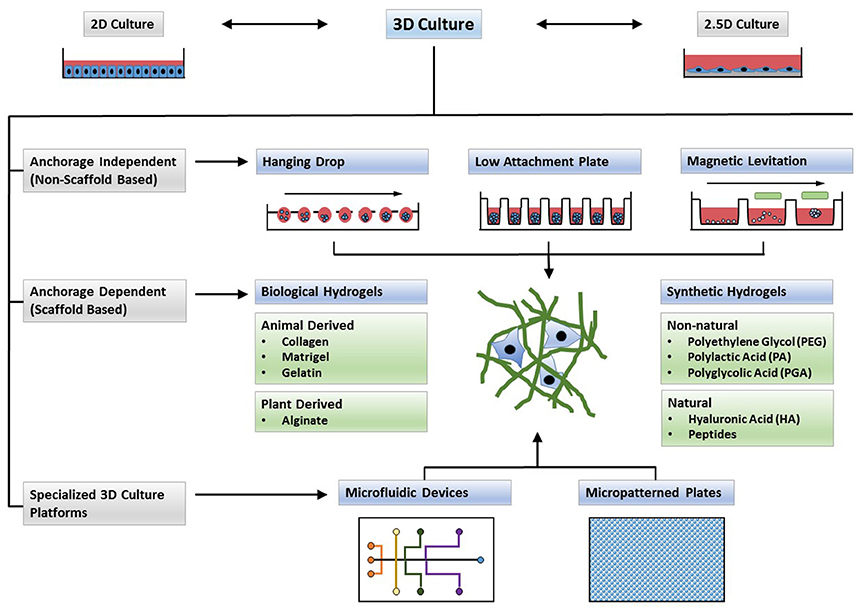
Scaffold dependent 3D cell cultures rely on physical support that ranges from mechanical surfaces to extra cellular matrix (ECM) like structures on which cells are cultivated to aggregate, divide and migrate. The physical and chemical properties of the scaffold will influence the cellular structure and characteristics. In synthetic scaffolds, hormones and growth factors necessary for the growth of a specific cell phenotype can be embedded within. Therefore the choice of scaffold, whether biological or synthetic, the physical (porosity, stiffness and stability under culture conditions) and chemical properties (compatibility) of the scaffold must be taken into account, to obtain a viable culture which can subsequently be used for high throughput drug screens (1).
Synthetic and Biological Hydrogels
Hydrogels are networks produced from dilute polymer chains with given structure and properties, obtained either by intermolecular or interfibrillar crosslinks. Biological hydrogels from natural sources such as collagen, fibrin or Matrigel are biocompatible, with natural adhesive properties and able to support many physiological cell functions. This results in increased cell viability, controlled proliferation or differentiation, and often a cell phenotype typically observed in vivo. Modification of the type of hydrogel used, and its concentrations is often used to observe cancer cell response to therapeutic measures that includes chemotherapy, immunotherapy and radiation (2,10).
However, most commonly used biological scaffolds such as collagen and Matrigel are available in liquid form but needs to be handled at cold temperatures to avoid premature gelation. This requirement for low temperatures limits its use in liquid handling equipment used for high-throughput screens in drug discovery (10).
Well designed synthetic hydrogels can serve as an excellent platform to use in 3D cell cultures. They can effectively represent ECM like properties, include proteolytic sites and growth factors. They can be easily fine tuned to achieve desired physical and chemical properties to promote the growth of a particular cellular phenotype. Additionally synthetic polymers have the added advantage of being comparatively inexpensive, relatively inert, and reproducible, thus enabling the acquisition of consistent and reliable results. Recent advances in material sciences have generated small peptide-based hydrogels as a new 3D cell culture scaffold that is amenable to drug discovery studies. These hydrogels are highly versatile and material properties can be modulated by substituting amino acids, either by extending or shortening the peptide sequence (3).
Common synthetic peptides effectively used for 3D culture are: the yeast-derived peptides EAK16 and RADA16 (4), the peptides Fmoc-FF (Fluorenylmethoxycarbonyl-diphenylalanine) (5) and Fmoc-RGD (Fluorenylmethoxycarbonyl arginine–glycine–aspartic acid) (6). A single acid substitution derivative of the MAX1 peptide, MAX8 with was shown to be compatible with liquid handling equipment making it suitable for high-throughput drug discovery (7).
Organoids
In the recent years organoid technology has rapidly evolved to be a 3D culture technique that results in self organising, self renewing, 3D cells derived from primary tissues, embryonic stem cells or induced pluripotent stem cells, that contain similar structure and function as the tissue of origin (8). Human cell derived organoids has paved the way for physiologically relevant models to study human disease development and progression. Advanced 3D organoid models further proves to be an effective platform for drug development, that is cost effective, with precise human disease models, that cannot be recreated in animal models (9).
The most common application of organoid cultures is transcriptome profiling, with evolving technologies compatible for high throughput drug screening. Kidney organoids have been studied in toxicity screens, along with a cystic fibrosis model developed for drug screens. In the recent years intestinal stem cells niche co-cultures has been developed for high throughput drug screens. Additionally, tumeroids (oranoids developed from patient specific cancer tissues) have provided a more representative 3D culture platform for personalised drug development (10).
References
- Caliari, S. R., and Burdick, J. A. (2016). A practical guide to hydrogels for cell culture. Nat. Methods
13, 405–414. - Worthington, P., Pochan, D. J., and Langhans, S. A. (2015). Peptide hydrogels – versatile matrices for
3D cell culture in cancer medicine. Front. Oncol. 5:92. doi: 10.3389/fonc.2015.00092Worthington, P.,
Pochan, D. J., and Langhans, S. A. (2015). Peptide hydrogels – versatile matrices for 3D cell culture in
cancer medicine. Front. Oncol. 5:92. - Zhang, Y. S., and Khademhosseini, A. (2017). Advances in engineering hydrogels. Science
356:eaaf3627. - Guilbaud, J. B., Vey, E., Boothroyd, S., Smith, A. M., Ulijn, R. V., Saiani, A., et al. (2010). Enzymatic
catalyzed synthesis and triggered gelation of ionic peptides. Langmuir 26, 11297–11303. - Zhou, M., Smith, A. M., Das, A. K., Hodson, N. W., Collins, R. F., Ulijn, R. V., et al. (2009). Selfassembled peptide-based hydrogels as scaffolds for anchorage-dependent cells. Biomaterials 30, 2523–2530.
- Worthington, P., Drake, K. M., Li, Z., Napper, A. D., Pochan, D. J., and Langhans, S. A. (2017). Betahairpin hydrogels as scaffolds for high-throughput drug discovery in three-dimensional cell culture. Anal. Biochem. 535, 25–34.
- Simian, M., and Bissell, M. J. (2017). Organoids: a historical perspective of thinking in three
dimensions. J. Cell Biol. 216, 31–40. - Dutta, D., Heo, I., and Clevers, H. (2017). Disease modeling in stem cell-derived 3D organoid systems. Trends Mol. Med. 23, 393–410.
- Langhans SA (2018) Three-Dimensional in Vitro Cell Culture Models in Drug Discovery and Drug
Repositioning. Front. Pharmacol. 9:6.
