Organ-On-a- Chip (OoC) Models of Colorectal Cancers
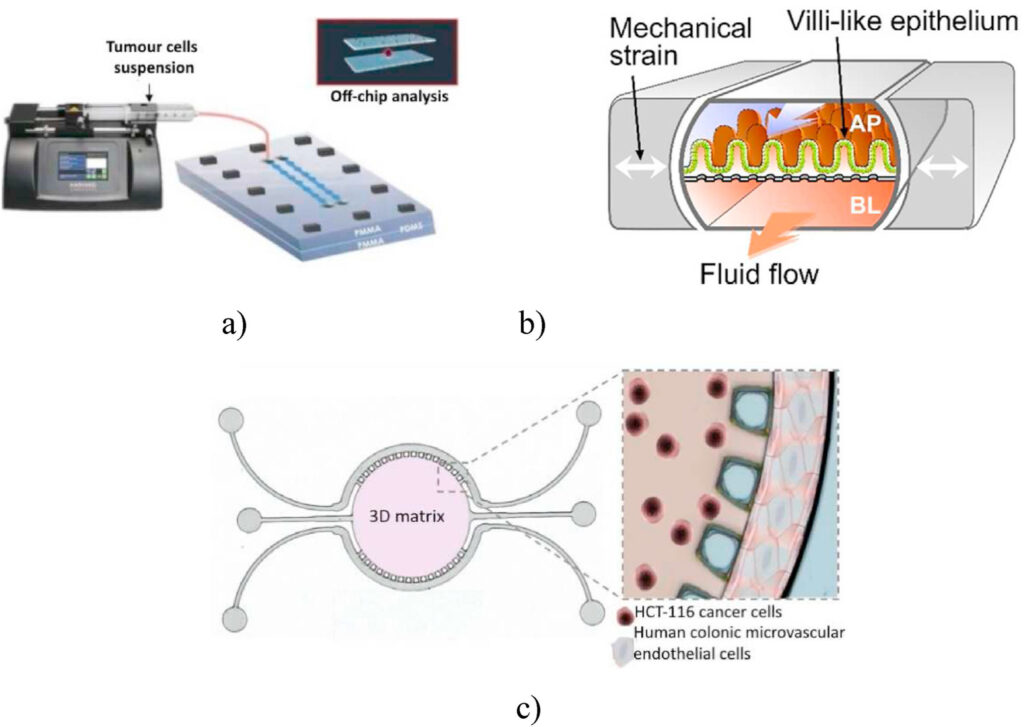
Colorectal cancer remains the second highest cause of death among all cancer-related mortalities. This is primarily due to tumor metastases and the inefficiency of available chemotherapeutic treatments (1,2). OoC models of colorectal cancers have been developed, however they are less common and have less success compared to OoC models of other types of cancers due to the challenges faced in replicating the complexity of the intestinal tissue architecture.
2D cell cultures were found inadequate in recreating some of the intestinal morphogenesis. The inefficiency of in vitro cell culture models causes lack of progress in understanding the disease progression and consequently in developing effective treatments. As a result, 3D cell culture models such as gut-on-a-chip models have emerged as the preferred platform for pathophysiological studies and drug screens (2-5).
Advances in gut-on-a-chip models to study disease progression
In an attempt to improve the quality of in vitro research models, 3D spheroids were produced in microwells within a gut-on-a-chip and connected to microchannels. These OoC was reversibly sealed permitting comprehensive evaluation of drug screens via scanning electron microscopy. OoC models developed from human primary intestinal organoids obtained from intestinal tissues harvested from a healthy individual also proved effectively in pathophysiological studies (2,6).
Gut-on-a-Chip models to develop potential treatments
Gut-on-a-chip devices have also been used to develop novel hydrogels as a treatment option to close gastrointestinal fistulas, which is a common complication of tumor growth and therapeutic treatments. The cells being cultured on these hydrogels expressed molecules involved in preventing the entry of gut bacteria and in the restoration of gut homeostasis (7,8). Researchers have also attempted to replicate the mucus layer by developing colon organoids from colon resections or biopsy specimens. The results were promising, as OoCs seemed to encourage the differentiation and formation of a mucus bilayer similar to the one found in in vivo conditions (9). A more exciting colorectal tumor–on-a-chip device consisted of tumor cells cultivated on Matrigel supplemented with vascular endothelial growth factor (VEGF). The lateral channels of the device were cultured with human colonic microvascular endothelial cells to establish a perfusable side channel with the lumen of the cells. This model can be employed for studying gene expression and offers the potential for patient-specific therapeutic treatments (10). Gut spheroids are indeed less common but they are essential to study normal cellular physiology and intestinal tumor progression. To this effect, more technological advancements are needed to produce a reliable gut-on-a-chip model that can mimic the complex in vivo architecture of intestinal tissues, thus yielding more clinically relevant data for drug development.
References
1. J. Komen, E.Y. Westerbeek, R.W. Kolkman, J. Roesthuis, C. Lievens, A. Van Den Berg, A.D. Van Der Meer, Controlled pharmacokinetic anti-cancer drug concentration profiles lead to growth inhibition of colorectal cancer cells in a microfluidic device, Lab Chip 20 (2020) 3167–3178.
2. Carvalho V, Bañobre-López M, Minas G, Senhorinha F.C.F. Teixeira, Lima R, Rodrigues R. The integration of spheroids and organoids into organ-on-a-chip platforms for tumour research: A review. Bioprinting, Volume 27 (2022) e00224.
3. L.C. Delon, Z. Guo, A. Oszmiana, C.C. Chien, R. Gibson, C. Prestidge, B. Thierry,A systematic investigation of the effect of the fluid shear stress on Caco-2 cells towards the optimization of epithelial organ-on-chip models, Biomaterials 225 (2019) 119521.
4. M.H. Kim, D. Kim, J.H. Sung, A Gut-Brain Axis-on-a-Chip for studying transport across epithelial and endothelial barriers, J. Ind. Eng. Chem. 101 (2021)126–134.
5. W. Shin, C.D. Hinojosa, D.E. Ingber, H.J. Kim, Human intestinal morphogenesis controlled by transepithelial morphogen gradient and flow-dependent physical cues in a microengineered gut-on-a-chip, iScience 15 (2019) 391–406.
6. J. Huang, Z. Li, Q. Hu, G. Chen, Y. Ren, X. Wu, J. Ren, Bioinspired anti-digestive hydrogels selected by a simulated gut microfluidic chip for closing gastrointestinal fistula, iScience 8 (2018) 40–48.
7. S.H. Tirumani, J.C. Baez, J.P. Jagannathan, A.B. Shinagare, N.H. Ramaiya, Tumor-bowel fistula: what radiologists should know, Abdom. Imag. 38 (2013) 1014–1023.
8. A. Sontheimer-Phelps, D.B. Chou, A. Tovaglieri, T.C. Ferrante, T. Duckworth, C. Fadel, V. Frismantas, A.D. Sutherland, S. Jalili-Firoozinezhad, M. Kasendra, E. Stas, J.C. Weaver, C.A. Richmond, O. Levy, R. Prantil-Baun, D.T. Breault, D. E. Ingber, Human colon-on-a-chip enables continuous in vitro analysis of colon mucus layer accumulation and physiology, Cmgh 9 (2020) 507–526.
9. M.R. Carvalho, D. Barata, L.M. Teixeira, S. Giselbrecht, R.L. Reis, J.M. Oliveira, R. Truckenmüller, P. Habibovic, Colorectal tumor-on-a-chip system: a 3D tool for precision onco-nanomedicine, Sci. Adv. 5 (2019).
