Modeling Infectious Diseases In 3D Human Organoids
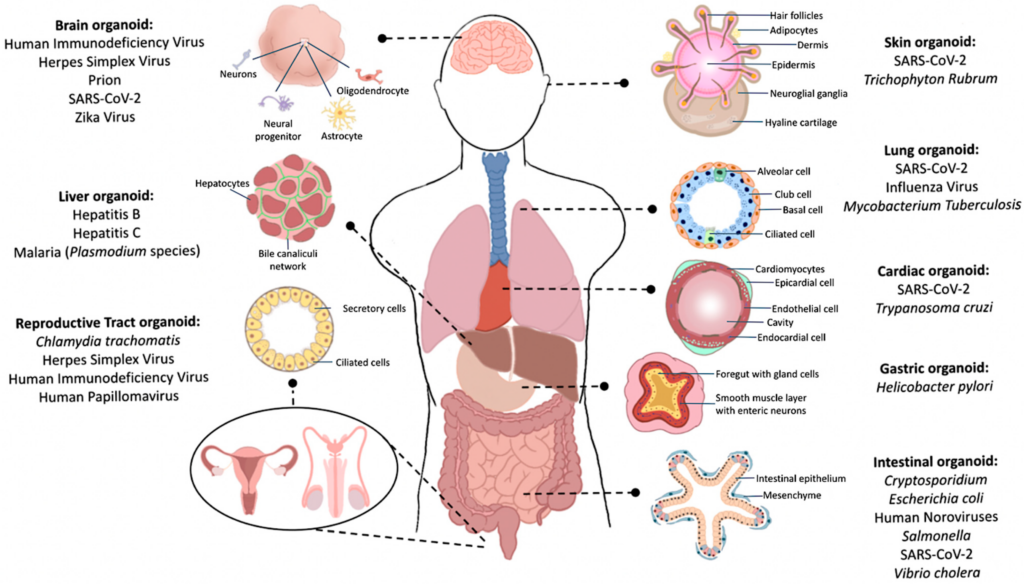
Organoids are 3D complex cluster of cells, derived from either stem cells or organ specific progenitors. When these cells are provided with an external scaffold like Matrigel, they form a complex cluster of cells that later differentiate to from a miniature version of an organ, able to mimic in vivo-like cellular architecture and function (1,2). Currently, most organoids can be categorized as tissue organoids or stem cell-derived organoids (3).
Stem cell-derived and tissue organoids
Stem cell-derived organoids can be generated from pluripotent embryonic stem (ES) cells, induced pluripotent stem (iPS) cells, or organ-specific adult stem cells (aSCs) (4). Tissue organoids rely on the ability of epithelial cells to self-assemble and form tissue-like structures and of growth factors to mimic the in vivo cellular environment.
Organoid technology is compatible with a wide range of tissues, organoids are stable over long periods and show a comprehensive 3D tissue architecture, making them the popular choice for many biological and clinical applications. To date a broad array of organoids have been established from several human tissues such as thyroid, pancreas, liver, stomach, cerebral cortex, intestine, kidney, lung, and retina (2).
Infectious disease studies
In the past decade organoid 3D culture models have been used for infectious disease studies. For example, brain organoids were used to investigate the impact of Zika Virus on human brain development, and the correlation between Zika Virus and microcephaly (5). Intestinal organoid models have been developed to study host-pathogen interactions in rotavirus (6), and norovirus (7). The impact of Helicobacter pylori on the stomach has been investigated by injecting bacteria into the lumen of gastric organoids (8).
More recently, hepatic organoids have been used to investigate the impact of COVID-19. These hepatic organoids were derived from human pluripotent stem cells and showed a high vulnerability to SARS-CoV 2 infection, making it an excellent model to investigate the pathogenicity of the virus and the resulting cellular response (9,10).
Organoids are a reliable and versatile platform, providing better insights into the in vivo state of cells and organs. Thanks to the rapid advancement of organoid technology which makes organoid generation easy to obtain, rapid and reproducible, organoids has a bright future ahead within infectious disease research.
References
1. Clevers H. (2016). Modeling Development and Disease with Organoids. Cell. 165 (7), 1586–1597.
2. Cacciamali A, Villa R and Dotti S (2022) 3D Cell Cultures: Evolution of an Ancient Tool for New Applications. Front. Physiol. 13:836480.
3. Huch M., Koo B.-K. (2015). Modeling Mouse and Human Development Using Organoid Cultures. Development 142 (18), 3113–3125.
4. Lancaster M. A., Knoblich J. A. (2014). Organogenesis in a Dish: Modeling Development and Disease Using Organoid Technologies. Science 345 (6194).
5. Wen Z., Song H., Ming G.-l. (2017). How Does Zika Virus Cause Microcephaly? Genes. Dev. 31 (9), 849–861.
6. Yin Y., Bijvelds M., Dang W., Xu L., van der Eijk A. A., Knipping K., et al. (2015). Modeling rotavirus Infection and Antiviral Therapy Using Primary Intestinal Organoids. Antiviral Res. 123, 120–131.
7. Ettayebi K., Crawford S. E., Murakami K., Broughman J. R., Karandikar U., Tenge V. R., et al. (2016). Replication of Human Noroviruses in Stem Cell-Derived Human Enteroids. Science 353 (6306), 1387–1393.
8. Bartfeld S., Bayram T., van de Wetering M., Huch M., Begthel H., Kujala P., et al. (2015). In Vitro expansion of Human Gastric Epithelial Stem Cells and Their Responses to Bacterial Infection. Gastroenterology 148 (1), 126–136.
9. Yang L., Han Y., Nilsson-Payant B. E., Gupta V., Wang P., Duan X., et al. (2020). A Human Pluripotent Stem Cell-Based Platform to Study SARS-CoV-2 Tropism and Model Virus Infection in Human Cells and Organoids. Cell. Stem Cell. 27 (1), 125–136.
10. Zhu L., She Z.-G., Cheng X., Qin J.-J., Zhang X.-J., Cai J., et al. (2020). Association of Blood Glucose Control and Outcomes in Patients with COVID-19 and Pre-existing Type 2 Diabetes. Cell. Metab. 31 (6), 1068–1077.
