Introducing nucleic acids – Transfection of eukaryotic cells
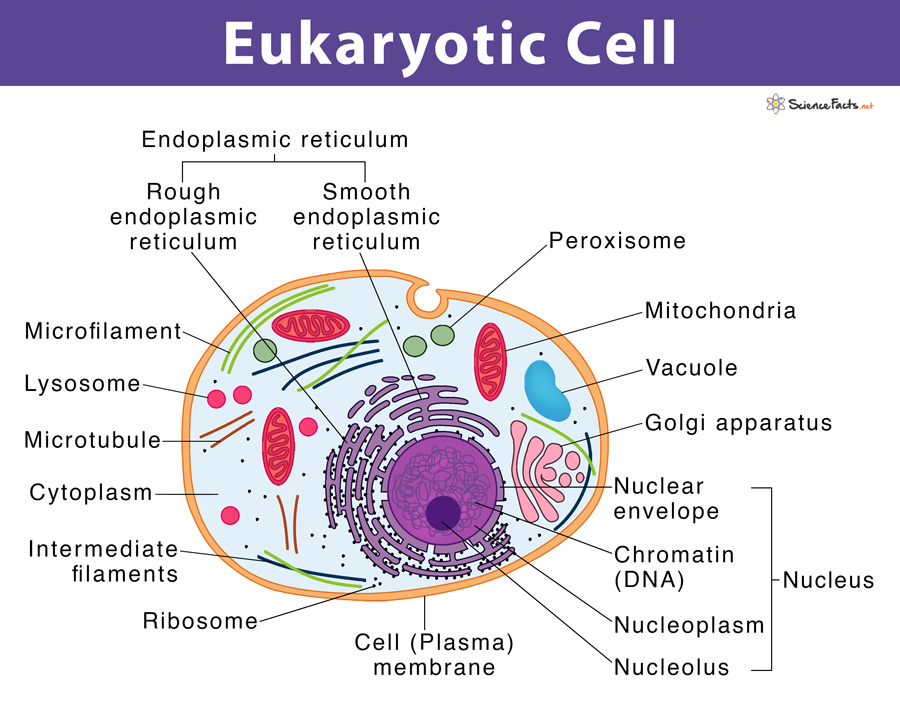
Transfection is a widely used technique to introduce foreign nucleic acids into eukaryotic cells. It is an essential tool enabling studies on gene function and gene products in cells. Advances in research techniques have enabled the transfection of various types of nucleic acids into mammalian cells including – DNA (deoxyribonucleic acid), RNA (ribonucleic acid), as well as some other small non-coding RNA such as siRNA, shRNA, and miRNA (1).
Transfection method in eukaryotic system: classification
Transfection can be classified into two types: stable and transient transfection. Stable transfection refers to sustained long-term expression of a transgene by integrating foreign DNA into the host nuclear genome. Whereas transient transfection does not require integrating nucleic acids into the host genome (2).
The choice of optimal transfection method depends on the type and origin of cells and the kind of nucleic acid to be introduced. There are various strategies for introducing nucleic acids into cells that utilize a variety of biological, chemical, and physical methods (3).
The most used method is the biological method, where nucleic acids are delivered to cells via viruses. The chemical methods are techniques that catalyse DNA cross-membrane transport using calcium phosphate or polycations. The physical transfection approaches include microinjection, optical transfection, and electroporation (4).
Each method has its own advantages and disadvantages, and the optimum method depends on experimental design and aim. Therefore, a wide variation is observed with respect to transfection efficiency, cell toxicity, effects on normal physiology of cells and the level of gene expression. However,, these major advances in transfection methods have made the introduction of defined nucleic acid sequences more effective, and this holds great promise for further use in gene therapy (4).
REFERENCES :
1. Borawski, J., Lindeman, A., Buxton, F., Labow, M., and Gaither, L. A. (2007). Optimization procedure for small interfering RNA transfection in a 384-well format. J. Biomol. Screen. 12, 546–559. doi: 10.1177/1087057107300172
2. Kim, T. K., and Eberwine, J. H. (2010). Mammalian cell transfection: the present and the future. Anal. Bioanal. Chem. 397, 3173–3178. doi: 10.1007/s00216-010-3821-6
3. Fus-Kujawa, A., Teper, P., Botor, M., Klarzyńska, K., Sieroń, Ł, Verbelen, B., et al. (2021). Functional star polymers as reagents for efficient nucleic acids delivery into HT-1080 cells. Int. J. Polym. Mater. 70, 356–370. doi: 10.1080/00914037.2020.1716227
4. Agnieszka Fus-Kujawa1, Pawel Prus1,2, Karolina Bajdak-Rusinek3, Paulina Teper4, Katarzyna Gawron1, Agnieszka Kowalczuk4 and Aleksander L. Sieron1* An Overview of Methods and Tools for Transfection of Eukaryotic Cells in vitro, REVIEW article, Front. Bioeng. Biotechnol., 20 July 2021 Sec. Preclinical Cell and Gene Therapy
