The Importance Of The Tumor Microenvironment In Cancer Research
Tumors derive from an abnormal growth of cells that occurs when cells continue to divide more than they should or when they do not enter programmed cell death. Tumors can be either benign or malignant. Malignant tumors are of most concern as they can grow uncontrollably and invade nearby tissues or other parts of the body, leading to severe diseases that often become fatal when left untreated.
Despite the significant investments being made in cancer research, less than 5% of the clinical interventions actually make it to the bedside. This is primarily due to the inability of available pre-clinical research models (2D cell cultures and animal models) to accurately represent the complex human cancer microenvironment (1).
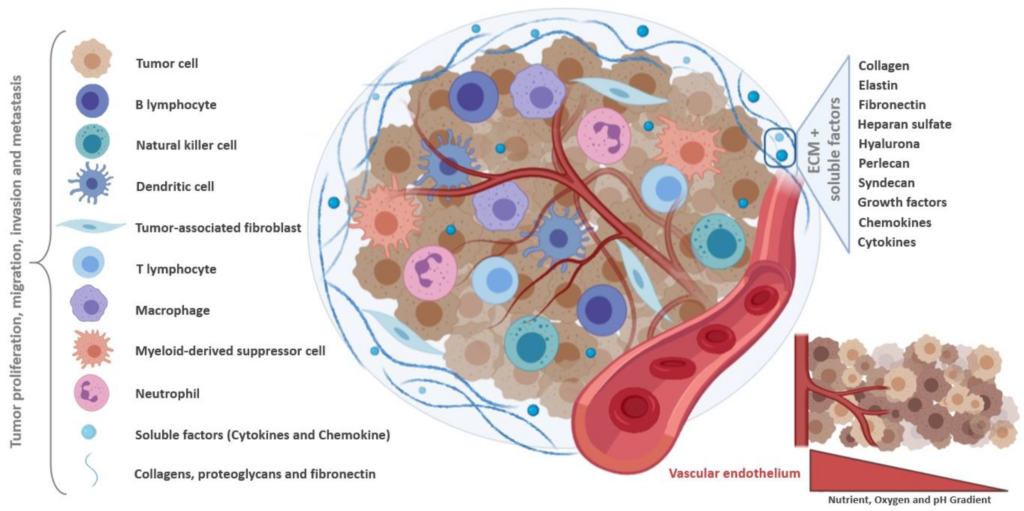
Components that make up the tumor microenvironment (TME)
The TME is composed of heterogeneous populations of tumor cells, along with the extracellular matrix (ECM) and other tumor associated cells such as fibroblasts, endothelial cells, adipocytes and immune cells. These immune cells include tumor-associated macrophages (TAM), which have phenotypic resemblance to M1 inflammatory macrophages that are involved in phagocytosis and cytotoxic lysis, and M2 inflammatory macrophages that mediate tissue repair (2).
The cytokines and the hypoxic state found within the TME promote the M2 phenotype, which subsequently aids in tissue repair, as well as tumor invasion and progression (3,4). TAMs make up approximately 50% of the tumor mass, and they often correlate with poor prognosis in most cancer types. Fibroblasts also play a significant role in the TME, in tumor cell invasion and in regulating changes to tumor growth and immune microenvironment through ECM remodeling and production of soluble factors (5,6).
The ECM within the TME offers structural support for the cells, and contains fibrous proteins such as collagens, multi-adhesive proteins, glycosaminoglycans, proteoglycans, growth factors and other secreted proteins. The cell-cell and cell-ECM communication is vital in determining tumor growth, progression and metastasis (7-9).
Molecular support for the tumor cells is provided by several factors present within the TME such as TGF-β, cytokines (IL-10 and IL-1β), VEGF, platelet-derived growth factor (PDGF), FGF, angiopoietins, Bv8/PROK2, and hypoxia-inducible factor (HIF) (10,11).
Cancer cells can modify their microenvironment to ensure survival. Tumor cells can utilize fibroblasts to get growth factors necessary for cell growth and proliferation. Interaction of tumor cells with neighboring endothelial cells results in release of soluble factors that mediates angiogenic processes. Tumor cells are also able to evade the natural immune-mediated response against them using the components of the TME. Loss of tumor antigen expression prevents tumor cells from being recognized by the immune system, and production of immunosuppressive cytokines guards them against cytotoxic lysis by immune cells.
All such strategies force the local TME to become immunosuppressive overall, causing the normal immune cells to become pro tumor in their response and to facilitate the overall tumor growth and progression (12-14). Therefore, the complex nature of TME and its role in tumor growth and progression need to be taken into consideration when designing clinical interventions that effectively target tumor cells.
References
- Pinto B, Henriques AC, Silva PMA, Bousbaa H. Three-Dimensional Spheroids as In Vitro Preclinical Models for Cancer Research. Pharmaceutics. 2020; 12(12):1186.
- Zhou, J.; Tang, Z.; Gao, S.; Li, C.; Feng, Y.; Zhou, X. Tumor-Associated Macrophages: Recent Insights and Therapies. Front. Oncol. 2020, 10, 188
- Wei, C.; Yang, C.; Wang, S.; Shi, D.; Zhang, C.; Lin, X.; Liu, Q.; Dou, R.; Xiong, B. Crosstalk between cancer cells and tumor associated macrophages is required for mesenchymal circulating tumor cell-mediated colorectal cancer metastasis. Mol. Cancer 2019, 18, 64.
- Lin, Y.; Xu, J.; Lan, H. Tumor-associated macrophages in tumor metastasis: Biological roles and clinical therapeutic applications. J. Hematol. Oncol. 2019, 12, 76
- Garufi, A.; Traversi, G.; Cirone, M.; D’Orazi, G. HIPK2 role in the tumor-host interaction, Impact on fibroblasts transdifferentiation CAF-like. IUBMB Life 2019, 71, 2055–2061
- Sahai, E.; Astsaturov, I.; Cukierman, E.; DeNardo, D.G.; Egeblad, M.; Evans, R.M.; Fearon, D.; Greten, F.R.;Hingorani, S.R.; Hunter, T.; et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat. Rev. Cancer 2020, 20, 174–186.
- Wagner, J.; Rapsomaniki, M.A.; Chevrier, S.; Anzeneder, T.; Langwieder, C.; Dykgers, A.; Rees, M.; Ramaswamy, A.; Muenst, S.; Soysal, S.D.; et al. A Single-Cell Atlas of the Tumor and Immune Ecosystem of Human Breast Cancer. Cell 2019, 177, 1330–1345.
- Reina-Campos, M.; Moscat, J.; Diaz-Meco, M. Metabolism shapes the tumor microenvironment. Curr. Opin. Cell Biol. 2017, 48, 47–53.
- Gonzalez, H.; Hagerling, C.; Werb, Z. Roles of the immune system in cancer: From tumor initiation to metastatic progression. Genes Dev. 2018, 32, 1267–1284.
- Zhang, J.; Zhang, Q.; Lou, Y.; Fu, Q.; Chen, Q.; Wei, T.; Yang, J.; Tang, J.; Wang, J.; Chen, Y.; et al. Hypoxia-inducible factor-1α/interleukin-1β signaling enhances hepatoma epithelial-mesenchymal transition through macrophages in a hypoxic-inflammatory microenvironment. Hepatology 2018, 67, 1872–1889.
- Kashyap, A.S.; Schmittnaegel, M.; Rigamonti, N.; Pais-Ferreira, D.; Mueller, P.; Buchi, M.; Ooi, C.H.; Kreuzaler, M.; Hirschmann, P.; Guichard, A.; et al. Optimized antiangiogenic reprogramming of the tumor microenvironment potentiates CD40 immunotherapy. Proc. Natl. Acad. Sci. USA 2020, 117, 541–551.
- Protopsaltis, N.J.; Liang, W.; Nudleman, E.; Ferrara, N. Interleukin-22 promotes tumor angiogenesis. Angiogenesis 2019, 22, 311–323.
- O’Donnell, J.S.; Teng, M.W.L.; Smyth, M.J. Cancer immunoediting and resistance to T cell-based immunotherapy. Nat. Rev. Clin. Oncol. 2019, 16, 151–167.
- Domschke, C.; Schneeweiss, A.; Stefanovic, S.; Wallwiener, M.; Heil, J.; Rom, J.; Sohn, C.; Beckhove, P.; Schuetz, F. Cellular Immune Responses and Immune Escape Mechanisms in Breast Cancer, Determinants of Immunotherapy. Breast Care 2016, 11, 102–107.
