The Shared Characteristics Of Spheroids And Tumors
Since its introduction, 3D cell culture has revolutionized cancer research by providing an efficient and modifiable research platform, that recapitulates in vivo like cell structure, function, gene expression and tissue microenvironment, all of which are important parameters for the drug discovery process. In addition to circumventing limitations posed by 2D monolayer cultures, 3D cultures also enable to reduce or ideally remove the use of animal models thus resolving ethical and economical concerns associated with animal model usage (1-4).
There are anchorage-dependent and independent 3D culturing methods. Among the most common and highly effective culturing techniques are spheroid culture methods.
Cancer spheroids: An ideal model for research
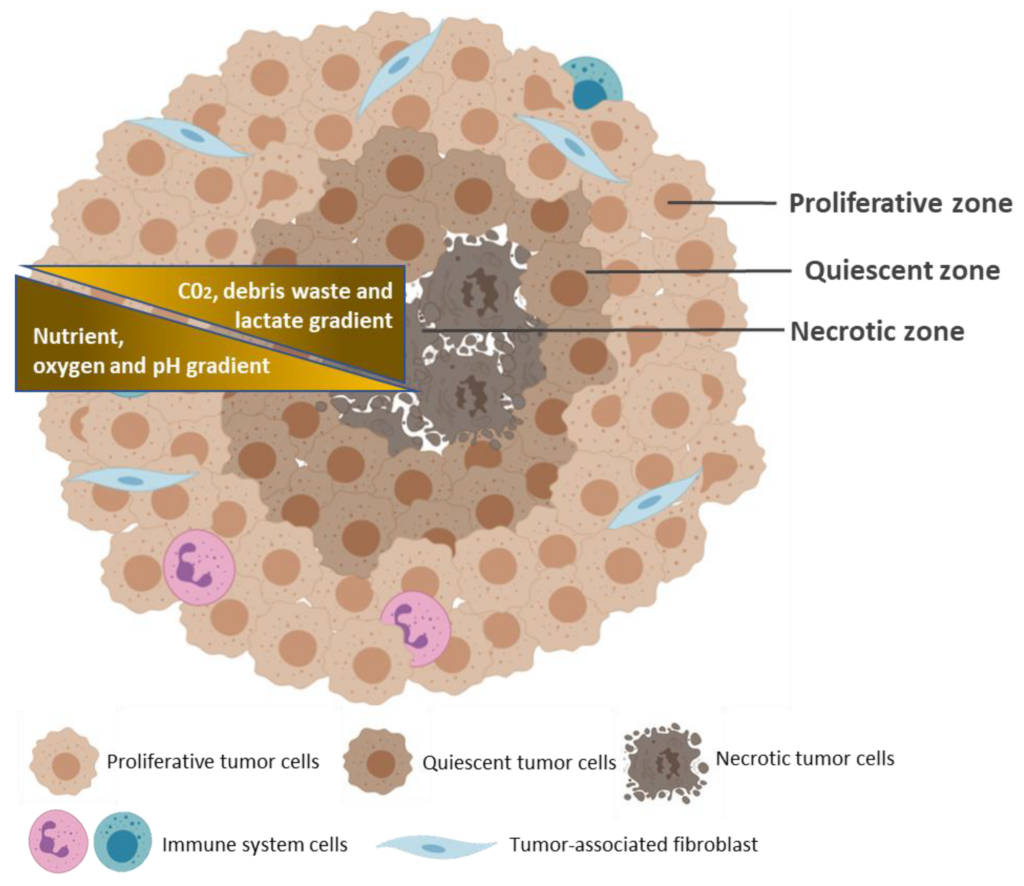
Like most self-aggregating cells, cancer cells can spontaneously assemble into spheroids, with in vivo-like cell-cell, and cell- ECM interactions (2). The 3D configuration of the spheroid allows for tumor cells to proliferate, aggregate and differentiate. Spheroids are typically 200 micrometers in diameter or more, and contain three concentric zones of heterogeneous cell populations.
There is an external zone of highly proliferating and migrating cells, a middle zone containing quiescent cells, and an internal zone of necrotic cells. These zones are determined by the availability of nutrients and oxygen from the outside to the center of the spheroids. The peripheral layer of cells is exposed to adequate amounts of oxygen and nutrients and therefore contains proliferating cells. The middle zone is exposed to limited amounts of nutrients and growth factors, and consequently contains quiescent cells. The lack of nutrient and oxygen, along with the accumulation of metabolic wastes, results in the necrotic inner cell layer (5-7).
Spheroid culturing systems are ideal for recapitulating the in vivo characteristics of tumor masses, the cell-cell and cell-ECM interactions, the nutrient and oxygen gradients, and the heterogeneous cell populations associated with cancer cells. Spheroid cultures are also able to mimic the gene expression and cell signaling pathways inherent to tumor cells, thus making it an efficient platform for studying cancer biology, and drug screening processes (8).
Ultra-low attachment plates (ULA): Making low cost, low tech, and high throughput compatible spheroid
ULA plates are among the scaffold free methods commonly employed to generate cancer spheroids. The surfaces of these plates are coated with special substrate/polymer rendering the surface cell repellant. This promotes the spontaneous aggregation of cells and spheroid formation. The wells of ultra-low attachment plates are also of a defined round, V, or conical shape, facilitating the formation of single spheroid with a consistently uniform size and shape (9,10).
The culture plates can be of varying formats (96 wells or 384 wells) and can be coated with special polymers such as commercially available BIOFLOATTM plates or with other substrates such as agar/agarose. This manner of tumor spheroid generation is high effective and efficient at generating large quantities of uniform spheroids in the same plate promoting reproducibility. Furthermore, they are relatively low cost, require simple tools and allows for monitoring of spheroid formation and growth. The size and quantity makes this method compatible for high throughput screening (8,11).
References
- Bissell, M.J. Architecture Is the Message, The role of extracellular matrix and 3-D structure in tissue-specific gene expression and breast cancer. Pezcoller Found J. 2007, 16, 2–17.
- Ravi, M.; Paramesh, V.; Kaviya, S.R.; Anuradha, E.; Solomon, F.P. 3D cell culture systems: Advantages and applications. J. Cell Physiol. 2015, 230, 16–26
- Białkowska, K.; Komorowski, P.; Bryszewska, M.; Miłowska, K. Spheroids as a Type of Three-Dimensional Cell Cultures-Examples of Methods of Preparation and the Most Important Application. Int. J. Mol. Sci. 2020, 21, 6225.
- Shehzad, A.; Ravinayagam, V.; AlRumaih, H.; Aljafary, M.; Almohazey, D.; Almofty, S.; Al-Rashid, N.A.; Al-Suhaimi, E.A. Application of Three-dimensional (3D) Tumor Cell Culture Systems and Mechanism of Drug Resistance. Curr. Pharm. Des. 2019, 25, 3599–3607.
- Baker, B.M.; Chen, C.S. Deconstructing the third dimension: How 3D culture microenvironments alter cellular cues. J. Cell Sci. 2012, 125, 3015–3024
- Hirschhaeuser, F.; Menne, H.; Dittfeld, C.; West, J.; Mueller-Klieser, W.; Kunz-Schughart, L.A. Multicellular tumor spheroids: An underestimated tool is catching up again. J. Biotechnol. 2010, 148, 3–15
- Longati, P.; Jia, X.; Eimer, J.; Wagman, A.; Witt, M.R.; Rehnmark, S.; Verbeke, C.; Toftgård, R.; Löhr, M.; Heuchel, R.L. 3D pancreatic carcinoma spheroids induce a matrix-rich, chemoresistant phenotype offering a better model for drug testing. BMC Cancer 2013, 13, 95
- Pinto B, Henriques AC, Silva PMA, Bousbaa H. Three-Dimensional Spheroids as In Vitro Preclinical Models for Cancer Research. Pharmaceutics. 2020; 12(12):1186
- Fang, Y.; Eglen, R.M. Three-Dimensional Cell Cultures in Drug Discovery and Development. SLAS Discov. 2017, 22, 456–472
- Gao, W.; Wu, D.; Wang, Y.; Wang, Z.; Zou, C.; Dai, Y.; Ng, C.F.; Teoh, J.Y.C.; Chan, F.L. Development of a novel and economical agar-based non-adherent three-dimensional culture method for enrichment of cancer stem-like cells. Stem Cell Res. Ther. 2018, 9, 243.
- https://facellitate.com/wp-content/uploads/Establishment-of-perfect-3D-spheroids-for-1.pdf
